






Welcome to the Summer 2012 edition of Winship Magazine.
Mark Saturday, Oct. 13, on your calendars!
We will be looking for you to join us that day at the Second Winship Win the Fight 5K Run/Walk!
Executive Director
Winship Cancer Institute
Walter J. Curran, Jr., MD Deputy Director
Fadlo R. Khuri, MD
Executive Administrator
Diane G. Cassels, MS
Director of Nursing
Deena Gilland, RN, MSN
Associate Vice President, Health Sciences Communications
Deputy Director
Fadlo R. Khuri, MD
Executive Administrator
Diane G. Cassels, MS
Director of Nursing
Deena Gilland, RN, MSN
Associate Vice President, Health Sciences Communications
Vincent J. Dollard, APR
Editor
Virginia L. Anderson
Graphic Designer
Linda Dobson
Photographer
Jack Kearse
Production Manager
Carol Pinto
Emory | Winship is published twice yearly by the Winship Cancer Institute Communications office for patients, families, staff, and friends. If you have story ideas or feedback, please contact virginia.l.anderson@emory.edu.
Website: cancer.emory.edu
It’s a special edition for us, as it marks the 75th anniversary of Robert Winship Woodruff’s extraordinary gift to Emory in 1937 to start a cancer clinic—only Mr. Woodruff didn’t want it to be called a “cancer clinic.” “People won’t come if you say the word ‘cancer’, he said when discussions were held about the name. He knew he wanted to name the clinic after his maternal grandfather, Mr. Robert Winship. From Winship, Woodruff inherited not only two-thirds of his name but also his love of the outdoors, his love of family, a deep-rooted love of Atlanta, and a desire to see it become one of the nation’s premier cities. When Mr. Woodruff’s mother, Emily Winship Woodruff, lay dying of cancer, he could not believe that nothing could be done for her. While he couldn’t save his mother, he decided to turn his grief into good for countless others who would be diagnosed with cancer. He made his first gift ever to Emory by giving $50,000 to start a clinic to treat cancer. A chief desire was that no one would have to leave the state of Georgia to receive the best cancer care possible. It’s a directive that the Robert Winship Clinic took seriously from the start, and it’s one that we at today’s Winship Cancer Institute of Emory University take seriously every day. We remain committed to Mr. Woodruff’s vision that no one should have to leave the state for cancer treatment. We’ve expanded the dream to encompass the vision that one day, no one will have to be treated for cancer anywhere because our understanding of cancer will have led to its prevention.
In this issue, you will read about progress we are making toward that vision in head and neck cancer, lung cancer, and other malignancies. And you will read about the second Winship Win the Fight 5K, scheduled for Oct. 13! We hope you start training now, and we look forward to seeing you there. Most of all, we thank you for your continued, generous support of Winship and the work that we do. Thank you for joining us in this fight!
Sincerely,
Walter J. Curran Jr., MD Executive Director, Winship Cancer Institute of Emory University
Winship Cancer Institute started with a love story. It is a story full of love for a woman named Emily Winship Woodruff and her family—and compassion and concern for all Georgians with cancer.

The head and neck cancer program takes teamwork to an entirely new level, hitting one of Georgia’s most prevalent cancers.
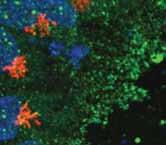

Survivorship means adapting to a new normal. Winship is there to help survivors stay well.
Good guys are focusing on the bad guys in lung cancer.
When Melanie Seymore married after treatment, she knew just what she wanted for wedding gifts — donations to Winship.
New research has provided leads for strategies against a mutated gene in some lung cancers.



years of caring
The bedrock value of Winship Cancer Institute is compassion. It only makes sense that it all began with a love story – one that has spanned generations and led to the remarkable advances Winship is leading today.
 By LYNNE ANDERSON
By LYNNE ANDERSON
The history of Winship Cancer Institute, which celebrates its 75th anniversary this year, actually starts with a love story that began in 1883.


The power of this love story has continued through the decades, into more than a century, and it lives at Winship today. Over the years, it inspired millions of dollars of philanthropy and immeasurable compassion. It resulted in thousands upon thousands of people’s lives being touched, many of them cured from the disease that claimed the woman at the heart of this story.
Her name was Emily Winship.
She was born in Atlanta in 1867, soon after the Civil War. Her father, Robert Winship, ran Winship Bro. Manufacturing. The company first made railroad ties and, later, Confederate artillery. Sherman put an end to both.

After the war, Winship Bro. Manufacturing was rebuilding, like the rest of Atlanta. By Emily’s teenage years, Winship Bro. Manufacturing had risen to become one of the nation’s largest foundries, occupying space in downtown Atlanta that is now near the site of the World Congress Center.
Robert Winship’s family lived in the fashionable Inman Park section of Atlanta. They happened to live next door to Ernest Woodruff’s sister, Annie, who had married Joel Hurt. That’s how Ernest Woodruff and Emily Winship met.
The Winships were devout Methodists. Robert Winship was a trustee of Emory College, and he was considered one of the city’s staunchest supporters and most generous philanthropists.
Ernest Woodruff helped his family run their flour milling business, Empire Mills, in Columbus, Georgia, a bustling river city. Empire made flour with such names as “King of Patents,” “Snow Flake,” and “Silver Leaf.”
By 1883, however, flour mills and cotton gins didn’t matter to Ernest and Emily. They were young and in love, and Ernest was on a mission to get the beautiful and kind Emily to return his affections.

father—that his love for her was true.
“If your father objects to our union, it is very unfortunate, but why should he be cool to the man who adores you,” he wrote in one letter. No doubt, it had to do with Emily leaving her loving family in Atlanta and moving to Columbus.
Ernest’s letters, written in a beautiful script that looks more like artwork than penmanship, are filled with the thrill of being in love.He finally won Emily’s father’s approval in 1884. He recognized the move to Columbus would be hard for Emily and her parents.

“I often think of your devoted parents and how grateful I should be to them and how much they will miss you,” Ernest wrote on Dec. 12, 1884, a few months before the couple married.
As the wedding date approached, he wrote:

“I hope that tomorrow will be the last day I shall ever address a letter to Miss Emie Winship.”

Finally, one day, a letter to her opens with these words, “My precious little wife.” It goes on
to say, “Every day I feel my unworthiness of such a good wife and only hope to be able to make you half as happy as you have made me.”
Robert Winship did indeed miss his oldest daughter. He wrote her soon after her move to Columbus, longing for a letter. “It is true that I have the benefit of all your sweet letters to Mamma and the rest, but sometimes you must address one to your Papa,” he wrote in June, 1885.

When the couple’s first child, a son, was born in 1889, they named him Robert Winship Woodruff in honor of Emily’s father.

Winship and his namesake grandson were very close. He taught the boy how to hunt and fish, pastimes that gave Woodruff great joy throughout his entire life.
The couple had a second son, whom they named after his father. They suffered a horrible loss when the young Ernest died of meningitis when he was two.

With Joel Hurt’s strong encouragement and

Robert Winship Woodruff donated $50,000 to Emory in 1937 to start the Winship Clinic.



generous invitation, Ernest Woodruff moved his family to Atlanta in 1893 to become involved in the running of several of Mr. Hurt’s enterprises, including what was to become the Trust Company of Georgia.
The couple faced another loss when Robert Winship died in 1899. His namesake grandson was almost 10. Emily and her children, particularly young Robert, were devastated.
Ernest became one of the city’s most successful businessmen, becoming president of the Trust Company of Georgia. The young romantic who had written that his only goal was to make Emily Winship happy now had a family to support—two other sons had been born to the couple—and empires to build. His greatest achievement may have been putting together the purchase of the CocaCola company.
The little boy the couple named after his grandfather later even surpassed his father in achievement, reaching peaks of success in American business unmatched by few in the 20th century.
His generosity to the city he grew up in is legendary. Robert Winship Woodruff’s philanthropy to Emory University alone—which totaled several hundred million dollars in his lifetime— would have been enough to secure his name forever in Atlanta.
Woodruff’s generosity to Emory all started in 1937, when his mother was


dying of breast cancer. Emily Winship Woodruff had had to travel to New York for treatment for her cancer.

It has been said that while Robert Woodruff had no children, he did have two sweethearts—his wife, Nell Hodgson Woodruff, and his mother. His love for both women was boundless. As his father had done, he too had written letters brimming with love to his mother, Emily.
“Give everybody my love, and keep a great deal for yourself,” he wrote in May, 1900, when he was 10.
When Emily and Ernest were traveling again in 1902, he wrote, “Hurry on back. I want to see you so badly. Your loving son, R.W. Woodruff.”
While at military school in 1907, he wrote his mother: “Your love prompts me to put forward my best efforts, and I trust I shall always be a comfort to you. Your loving son, R.W. Woodruff.”
While Woodruff was not an emotionally demonstrative man, his mother’s illness years later was devastating to him.
“I remember when his mother, whom he adored, got sick,” recalls Nell Hodgson Watt, niece of Nell Hodgson Woodruff and like a daughter to the couple. “He was just crushed at losing Mrs. Woodruff. He said, ‘I’m going to build a clinic just for cancer, so people will think Emory before they think of any other place.”
He and Nell sat by Emily’s bedside for days on end.
While Woodruff couldn’t stop cancer from robbing him of his beloved mother, he knew he could do something. He could keep her memory alive, honor his grandfather, and fight cancer at the same time.
And so with a $50,000 donation to Emory University, he started the Robert Winship Clinic. It was his first donation to
occupied the first floor of the east wing of Emory Hospital. Scarborough saw his first patient in 1937.
The clinic faced financial obstacles during its first several years, and Woodruff personally covered the shortfalls.

Through the years, Scarborough developed a reputation in Atlanta and across the nation for his vision—and informing the public about cancer.
who had become like a son to Robert Woodruff, had pancreatic cancer. He had only a short time to live.
His biggest concern was that patients would lose confidence in the Winship Clinic if its very heart and soul was dying of the disease himself.
The entire city mourned Scarborough’s death. During his nearly 30 years at Winship and Emory, he had earned a reputation as a cancer crusader and firstrate physician. He had attracted other outstanding physicians to join him, and it was one of his key hires, Sam Wilkins, who stepped into the very large shoes left by Elliott Scarborough.

Emory University. He then went to New York’s famed Memorial Hospital and hired fellow southerner and Harvard-educated Elliott Scarborough to run the clinic.
Robert Woodruff did not want the word “cancer” used in conjunction with the clinic. “Don’t call this a cancer clinic because people won’t come,” he told Scarborough. A news story from the time talks about X-ray and other “therapeutic machinery,” but no word is mentioned of cancer. Woodruff didn’t even want newspaper coverage; cancer was too taboo.
“I do not believe there should be a dedication at all,” he wrote about plans to mark the opening of the clinic, which
In 1949, Scarborough was named to the National Advisory Cancer Council. In 1954, long before the U.S. Surgeon General issued warnings about smoking, Scarborough publicly declared that he believed smoking caused lung cancer. In 1955, he was appointed to the board of directors of the American Cancer Society. Meanwhile, the Robert Winship Memorial Clinic was treating thousands of patients a year, saving lives and extending survival times. Cancer was no longer taboo.
With the operations running smoothly, the Winship Clinic was poised to help even more patients. In 1966, however, bad news came. Scarborough,
Under Scarborough’s direction, Winship Clinic had made a name for itself through its excellent treatment and compassionate care.
A 1956 letter to Woodruff from a patient shows her gratitude for the care she received at Winship Clinic:
“I have great praise for the doctors and nurses there especially Dr. Sam Wilkins …who has given me so much hope and courage…I only go for a checkup every six months now and I have everything to look forward to ahead, having 4 wonderful children and my dear husband and if my days are short, at least I have three years spared to me I never dreamed I could have and again I wish to say from the bottom of my heart Thanks for the Robert Winship Clinic.”
“Your love prompts me to put forward my best efforts, and I trust I shall always be a comfort to you,” - Woodruff to his mother.The first staff of the Robert Winship Clinic gathered for a group photo. Dr. Elliott Scarborough is seated on the right.

Robert W. Woodruff might not recognize the Winship Cancer Institute of today. After several decades of providing first-rate cancer care, Winship received an infusion of energy and resources in the late 1990s that, once again, had the hand and heart of Robert W. Woodruff in it. And, once again, the effects would be life-changing—and life-saving. They started in August, 1996, just after the Olympic Games in Atlanta. The Robert W. Woodruff Foundation Inc., the Joseph B. Whitehead Foundation, and the Lettie Pate Evans Foundation jointly designated a portion of their Coca-Cola stock to be set aside, with all dividend earnings to go to Emory’s Woodruff Health Sciences Center, which had been established in 1966 to focus on the missions of teaching, research, health care. and public service. The estimated value of the stock in 1996 was $295 million. At the time, The Chronicle of Higher Education suggested that this endowment was likely the largest single commitment ever made in American higher education.
The endowment was called the Robert W. Woodruff Health Sciences Fund, Inc. Its purpose was to support the Woodruff Health Sciences Center in meeting two primary goals: build the infrastructure of people and programs required for the Center to rapidly take its place among the nation’s leading academic health sciences centers, and to align the Center’s resources and momentum in the directions in which science, medicine, and patient care are expected to head in the 21st century.
In accordance with its original design, half of the annual income from the Fund, Inc. supports programs and facilities of the Winship Cancer Institute. The generous contributions from the Fund Inc. enabled Emory to build the 275,000 square-foot Winship Cancer Institute building.
Propelled in large part by this extraordinary gift, within a decade, Winship would: move into the state-of-the art building, attract

dozens of top–tier scientists from around the world; become one of an elite cadre of National Cancer Institute–designated cancer centers – and bring hope and healing to thousands of Georgians and residents of other states.

A big boost also occurred in 2000. It started with an idea hatched by Michael Johns, Emory chancellor and emeritus executive vice president for health affairs and the late Hamilton Jordan, former President Jimmy Carter’s White House Chief of Staff. Jordan had endured six bouts of cancer. Johns and Jordan put together a presentation to accelerate cancer prevention, research, and treatment in Georgia.
They took their presentation to then-Gov. Roy E. Barnes, who echoed the vision of Winship’s founder decades earlier when Woodruff declared that no resident of Georgia—not just Atlanta —should have to leave the state for the best cancer care. In the fall of 2000, Barnes announced the concept of the Georgia Cancer Coalition. In 2001, the Distinguished Cancer Clinicians and Scientists Program was initiated to attract renowned researchers to Georgia’s academic medical centers.

Jonathan Simons, a prostate cancer specialist recruited from Johns Hopkins, had become the new director of Winship in 2000. He was an integral part of the planning and construction of the new building that would allow bench-to-bedside research and treatment; it was his idea to have Winship’s core values embedded in the stairways.
After Simons’ departure, Brian Leyland-Jones was appointed director. He came to Atlanta from McGill University. Under his direction, Winship earned the prestigious National Cancer Institute Cancer Center Designation in 2009. With this designation, Winship joined the ranks of the most elite cancer centers in the country.
Leyland-Jones stepped down shortly after the designation. Walter J. Curran, Jr., chairman of Emory’s Department of Radiation Oncology, was then appointed Executive Director of Winship. Curran, who joined Emory in 2008 from Jefferson Medical College in Philadelphia, is internationally recognized in the treatment of patients with locally

advanced lung cancer and brain tumors. He continues to serve as the Lawrence W. Davis Chair of Radiation Oncology and is the only radiation oncologist in the country to lead an NCI-designated cancer center.




“I am very pleased with where we are, and I’m proud of our dedicated physicians, nurses, scientists, and everyone who works at Winship,” he says. “What makes me the happiest at the end of the day, however, is knowing that we are making a difference in people’s lives. We are lessening the burden of cancer in this state, which is what Robert Woodruff wanted his legacy to be. We are keeping his vision alive and doing the work he wanted people to do.”
Curran and others note that while many have contributed to the Winship Cancer Institute’s elite status, its current deputy director, Fadlo R. Khuri, has been a driving force of leadership and vision throughout Winship’s dynamic decade. Khuri came to Winship from MD Anderson in 2002, and he has recruited many of the nation’s brightest physicians and researchers to Winship.
“He also has played an integral role in keeping them here,” Curran says.
Khuri says that has more to do with Winship and its values than with his efforts.
“Winship has built a national reputation among physicians, nurses and researchers as being a place where hard work, integrity and collaboration are encouraged and appreciated,” says Khuri. “We have worked hard to build a culture I think Mr. Woodruff would be proud of—a culture in which accomplishments of the group are valued and celebrated more than individual accomplishments.”
Since the building dedication, Winship
Winship faculty are helping drive the national cancer conversation by publishing dozens of papers in high-impact journals and by their presence and leadership roles at the nation’s leading cancer symposia. A recent survey of National Institutes of Health grant funding in 2011 lists Curran, whose NIH research funding totaled $16,074,557 last year, as the only researcher in Georgia and the only NCI-designated cancer center director to be among the top 25 in NIH grant funding. And Khuri is editor of the prestigious, peer-reviewed journal Cancer, published by the American Cancer Society.
has hired close to 100 new clinical and laboratory investigators. Areas of excellence are difficult to point out because there are so many—drug development, patient care, nursing, supportive oncology and survivorship, a phase I clinical trial division, basic science, experts in areas ranging from bone marrow transplantation to new treatments for multiple myeloma, lung cancer biomarker identification, head and neck cancer, and triple negative breast cancer. Winship and Emory Healthcare recently announced an agreement to bring proton beam therapy to Atlanta, which will make Winship one of only a handful of cancer treatment centers in the country to offer this precise form of radiation therapy.

The value embedded on Winship’s bedrock level, where Curran keeps his office, is “Compassion.” On Valentine’s Day, survivor Deana “Rosie” Collins celebrated her last day of treatment on that tunnel-level floor. She was so happy that she painted a banner that read “Tunnel of Love; you are loved!” She passed out gifts to those who were part of her treatment plan. She handed out Valentinedecorated cupcakes to patients.
You have to think that Robert Winship Woodruff would be more than a little proud—and that his parents, Ernest and Emily, would be happy knowing how their love story still plays out and the immeasurable good that their son accomplished because of it.
The value embedded on the bedrock level of Winship is “Compassion.”
Winship’s head and neck cancer program defines multi-disciplinary care, and patients win from the teamwork—and hard work.

 By Lynne Anderson |
Photography by Jack Kearse
By Lynne Anderson |
Photography by Jack Kearse
The sun shows no signs of rising on this rainy February morning, and the streets are nearly bare. A drive down Clifton Road from the CDC to Winship takes two minutes flat, even after a red light or two snags you. Buses and shuttles are rare, and valet parking hasn’t opened.
So it’s a little surprising to walk into the boardroom on the fifth floor of Winship Cancer Institute to find a hive of activity and conversation.
About 50 doctors, nurses, and other members of Winship’s head and neck cancer team are wide awake and in full work mode, long before they will dash off to their clinics, operating rooms, labs, and offices to begin a full day’s work there.
It’s the head and neck program’s tumor board meeting, and it happens every Tuesday at 6:30 a.m. This team of experts comes from surgery, radiation and medical oncology, pathology, radiology, nutrition, nursing, social work and speech pathology. They will discuss every head and neck cancer patient seen in the past week before they leave.
While it may not be enough to rouse the average person out of bed at 5 a.m., those in attendance wouldn’t want to miss it. They know their collaboration makes a
difference in their patients’ lives and that it helps improve treatment.
“We present every single patient —including those seen at Emory Midtown and the VA Medical Center —to make the best decision for patient care,” says Dong Moon Shin, associate director of Winship and director of the head and neck cancer program at Winship. Even patients whose cancer may not appear to be life-threatening can have “major complications, major issues,” that physicians bring to the tumor board to discuss, explains Shin, a medical oncologist.
And this is not just any tumor board—it’s a working board of more than four dozen experts.
“There’s a lot of experience in that room,” says Jonathan Beitler, a radiation oncologist who specializes in head and neck cancers. “It’s a good way to see the same evidence at the same time. There are decades of surgical experience in the room. It’s a great way to provide patients the very best care.”
Preparation and organization are key, says Nabil Saba, a medical oncologist who specializes in head and neck cancers. “It’s become an example of what a multidisciplinary care program can be.”

Other tumor boards meet at Winship, and they all have patient care as the primary focus. The head and neck tumor board meeting may be one of the best examples, however, of the interdisciplinary work that sets Winship


“The whole thing is, you’ve got the Winship team, and you’ve embraced them, and they embrace you. They understand you as a person. It gives you so much confidence, which you need if you’re going to win this battle. ” – head and neck cancer survivor Greg Gregory.
apart—and helps ensure that patients receive the benefit of expertise from several disciplines.
Collaboration is becoming more and more key in unlocking cancer’s dark secrets. Winship is known for collaboration across disciplines and within departments; Winship’s deputy director, Fadlo R. Khuri, has said that a willingness to collaborate is a job requirement.
“The thing that we always remember is that we are here for the patients, not for ourselves,” says Khuri. “The head and neck cancer team is an outstanding example of brilliant scientists and clinicians coming together toward one common goal—helping our patients get well, with minimal side effects.”
It’s easy to see what Khuri is talking about when trying to interview and photograph members of the head and neck cancer team. Shin doesn’t want to leave anyone out and recites from memory the entire team and each of their responsibilities, excitedly talking about each one. When it’s time for a photo shoot, he wants to try to get everyone in the picture.
“This is one of the largest interdisciplinary tumor boards anywhere in the nation,” explains Shin. “Patients are receiving the best of care from the best of the experts. And that care is enhanced because we have so many committed members of the treatment team who come together with great knowledge and great compassion to make sure our patients are getting the best care possible.”
Winship’s head and neck cancer program stands out not only because of exceptional patient care but also because it is one of only five in the country to be designated a Head and Neck Cancer Specialized Program of Research Excellence, or SPORE. “We are very proud to have established such a prestigious program at Winship, and it’s also the only SPORE in the state of Georgia,” says Shin. SPORE grants are funded by the National Cancer Institute and bring with them about $2.5 million in research funding each year for five years. The research program is very comprehensive, consisting of four major programs, research shared resources, a career development program, and a developmental research program.
In a state and region where head and neck cancer rates are higher than the national average, the head and neck cancer program is a critical piece of Winship’s efforts.

Head and neck cancers include cancers of the mouth, lips, nasal cavity, sinuses, throat, tongue, salivary glands, and larynx. Risk factors include cigarette smoke and alcohol use, but many patients have never smoked or used alcohol. Winship treats about 750 newly diagnosed patients a year with the disease.
In recent years, the number of patients diagnosed with head and neck cancer had been falling. That was due, in some measure, to a decline in tobacco use. Now, a recent spike in certain types
“There’s evidence that shows that aggressive, supportive care improves outcomes. Instead of everyone going up for a pop fly, thinking someone else has it and the ball gets dropped, here, the ball doesn’t get dropped.” — otolaryngologist Amy Chen.
of head and neck cancers has been associated with the human papilloma virus-16, or HPV16. Many experts believe such cancers to be at epidemic levels (see sidebar).
Cancers of the head and neck region are difficult cancers. Treatment often limits a person’s ability to swallow, which in turn greatly affects his or her diet and nutrition. Surgery may change the appearance and function of a person’s face. Sometimes patients lose their sense of taste.
“Head and neck cancers affect a part of our body that we use as social creatures,” says Amy Chen, head and neck surgeon who started the tumor board in 2001.

“That’s what makes it so critical to have this collaborative care. This is a cancer that can devastate its patients. It affects their ability to be social, at a party, at a wedding, at dinner with friends. If you have surgery, everyone knows. It’s a very visible disease.”
Head and neck cancer patients also experience the effects of stigma associated with cancers that others sometimes believe the patients brought on themselves. Because tobacco and alcohol use are risk factors, head and neck cancer patients, like those with
lung cancer, sometimes “don’t have the sympathy” of other cancer patients, says Scott Kono, one of the newest members of the team.
“That’s one of the things that attracted me to the field, sort of the underdog aspect of these patients,” says Kono, who joined Winship about a year and a half ago.
Head and neck cancer patients need special care in that regard, Kono says, and they receive it at Winship. “There’s a high incidence of depression, and a high

suicide rate compared with other cancers,” Kono explains. That may be related to treatment intensity, a long recovery period, and people being cut off from their social network because of impediments to their speech and swallowing, and to effects on their appearance, says Kono. These issues too are discussed at tumor board.
Surgeons such as Chen are often the discussion leaders. As the team discusses each case, all in attendance pay close attention to Power Point slides being
displayed at the front of the room. CT, MRI, and PET scans, as well as photographs, are used. Surgeon Bill Grist stands at the front, presenting cases and asking and answering questions not only about best surgical interventions but also about the psycho-social issues patients are facing. The group does not move on to the next patient until all questions relating to the patient at hand are answered.
It’s not all doctors and nurses here; social worker Carol Rivera and speech pathologist Meryl Kaufman are also in attendance, guaranteeing patients a full spectrum of care. Kaufman explains that patients see speech pathologists before treatment even begins. Giving patients exercises not only helps them maintain muscle tone in their necks and throats but also helps them have some feeling of control as they go through treatment, Kaufman explains.
“Most hospitals don’t have diseasespecific tumor boards,” says Chen, “and the patient is comforted by knowing that we have such collaboration.”
Jim Stapleton, a retired Army colonel and head and neck cancer survivor, says he received “phenomenal care” at Winship. He is so appreciative that he brings pastries and coffee every single Tuesday morning from Highland Bakery, whose owner Stacey Eames even contributes by giving Jim a discounted rate because it’s Winship. Although Stapleton finished treatment nearly five years ago, he wants to do what he can to show his gratitude.
“These guys saved my life,” Stapleton says. “I’m impressed that they meet this early in the morning. It just shows their tremendous dedication.”
Patients do benefit medically from such coordinated care, Chen says.
“There’s evidence showing that aggressive, supportive care improves outcomes,” she says. “Instead of everyone going up for a pop fly, thinking someone else has it and the ball gets dropped, here the ball doesn’t get dropped.”
Greg Gregory of Atlanta was diagnosed

with Stage IV throat cancer in April, 2011. Before the year was out, he was healthy and traveling in Asia. Chen was his surgeon and also the “captain” of his team.
“You need somebody you can look up to and say ‘she’s in charge,’” Gregory says of Chen. “You want to salute her because you have so much confidence in her.”
As soon as he praises Chen, other names pop up, too. It’s as if the team is so interwoven that patients can’t think about one caregiver without wanting to praise all the rest of the team.
“Oh, and Dr. Saba (Nabil) is just a splendid man with a splendid staff,” Gregory says.
For his part, Saba simply says that it’s a privilege to be part of patients’ lives and to help them recover.
“It makes me feel so grateful that we do have such a system and such a program in place that we do make such a difference in people’s lives,” he says.
The exceptional team work is especially important in the treatment of head and neck cancers, explains Grist.
“This is a very labor-intensive cancer to treat,” says Grist, “and tumor board really has facilitated communication so that patients are getting the best care possible.”
Gregory summarizes it this way:
“The whole thing is, you’ve got the Winship team, and you’ve embraced them, and they embrace you. They understand you as a
Dong Moon Shin, associate director of Winship and director of the head and neck cancer program, says he is proud of his dedicated team of researchers, physicians, and nurses, who strive hard through teamwork “to make the best decision for patient care.”
person. It gives you so much confidence, which you need if you’re going to win this battle,” he says. “I don’t know how Winship accumulated all that talent in one place, but it’s pretty amazing.”
Winship’s head and neck cancer program has a strong research focus that supports its patient care. Because head and neck cancers can be challenging to treat and because no effective screening tool exists for them, the Winship approach to these cancers is aggressive.
For example, Winship, with National Cancer Institute SPORE grant support, has awarded 36 development awards to young investigators, says Shin.

The robust SPORE research team is involved in at least

a dozen clinical trials, including trials evaluating the best use of concurrent radiation and chemotherapy in those with high-risk squamous cell cancer, which accounts for a large percentage of head and neck tumors.
In addition, researchers are studying chemoprevention, nanotechnology therapies, and methods of blocking key pathways that cancer cells need to thrive.
Consider the work of Shin’s lab. Georgia Chen and Ruhul Amin are looking at whether pre-malignant head and neck lesions can be prevented from becoming head and neck cancer with the use of green tea polyphenon E.
Chen, Shin, Xianghong Peng, Xu Wang, Ximei Qian, Shuming Nie, and Beitler are studying the significance of circulating tumor cells detected by gold nanoparticles. These so-called CTCs can help doctors determine whether disease has metastasized.

Another important study is examining whether COX-2 inhibitors—the same type of drug that prevents inflammation-induced arthritis—in combination with EGFR tyrosine kinase inhibitors (TKIs) such as erlotinib—can prevent cancers from precancerous lesions. The approach makes sense. COX-2 expression is common in the upper aerodigestive tract, and COX-2 inhibitors already are being developed in chemoprevention for colon cancer.
Other trials are looking at what happens when COX-2 inhibitors are combined with EGFR-TKIs.
“The result (of the combination) is highly synergistic to inhibit cancer progression,” Shin says.
Another clinical trial is assessing the combination of EGFR-TKIs with green tea. That combination also works synergistically to inhibit tumor growth, Shin explains.
The SPORE grant also includes a pathology and statistics core. And the program features a strong career development program (CDP) and research development program (RDP) to nurture young investigators and developing science. The CDP program, under the direction of Winship Deputy Director Fadlo R. Khuri, gave 19 awards. The RDP, led
While patients with HPV16related cancers generally have better outcomes than patients without an HPV16 association, cancers caused by HPV16 are still a matter of great concern at Winship.
“It’s an epidemic,” says Dong Moon Shin.
Shin and others point to the growing numbers of patients with HPV16-associated head and neck cancer in their 30s and 40s without traditional risk factors for head and neck cancer.
HPV16 infection is generally caused by sexual contact, but one of the puzzling parts about the virus is that not all people who are infected with HPV16, which includes about 20 million American adults, will develop cancer.
Researchers do know that the virus can be latent for 10 years or more, Bill Grist explained recently at a survivors’ support group meeting. And they also know that having six or more oral sex partners in one’s lifetime increases a person’s risk of developing oropharyngeal cancer nine times. Compare that to the increased risk caused by drinking—2.5 times—and smoking—3 times—and it’s easy to understand the concern.
Winship researchers are trying to understand how HPV16 is involved with carcinogenesis and why some people develop cancer from it and others do not.
Because patients with HPV16-related cancer fare better in many cases than those with other kinds of head and neck cancer, Winship doctors are also conducting trials to determine whether some of those cases need as much treatment as others do.
Shin and others were very supportive of guidelines suggested last fall to vaccinate boys and girls with a vaccine that can prevent HPV16 infection. Winship experts suggest that parents of boys and girls talk to their pediatricians about the vaccine to protect against HPV16.
Dong, gave 17 awards over the past five years.
“Such pilot programs of research clearly help young investigators to devote themselves to perform meaningful research in head and neck cancer,” Shin says.
It’s 6:30 p.m. on another February day. Grist is the guest speaker at a head and neck cancer survivors’ support group, which surgeon Chen and nurse Arlene Kehir started years ago. Beitler is there too. The room is packed.
Grist presents information on the rising incidence of oropharyngeal cancers caused by HPV16. The rising incidence concerns Grist, Beitler, and other head and neck cancer doctors.

Both doctors express a sense of urgency in finding answers to why millions of people clear the virus every year and why others don’t. They also want to understand how cells become cancerous when infected by the virus. Yet another area of research at Winship on HPV16 focuses on treatment of these patients. Research and experience have shown that these patients fare better than other patients with head and neck cancer. So Winship is conducting and participating in clinical trials, explains Beitler, to study whether de-intensifying treatment in this patient group might work. For example, might patients do just as well with cetuximab, a monoclonal antibody without a range of side effects, as they do on standard therapy with cisplatin, which does have a range of significant side effects.
“You really have to study at night to keep up with the changes,” says Beitler, who also happens to be a flight surgeon in the National Guard (a colonel) and who also holds an MBA
Howard and Lynne Halpern, left and center, recently made a $2.5 million planned gift to honor their friend and physician Fadlo R. Khuri, right, and to support the development of new therapies for head and neck cancers.

(“The main thing I learned is that I care more about treating patients than making money,” he says).
Marcy Leamy is only one of several at the support group who talk about how that dedication has made a difference in their lives.
“Dr. Beitler saved my dad’s life,” says Leamy, whose father Alfred, a mathematician, recently underwent surgery for head and neck cancer. “My father is from Tampa, and they called up here and said, ‘We have a stage IV; can you see him?’”
Beitler did. Albert needed surgery right away. “He walked us up to the surgeon’s office that very minute,” Marcy recalls. “They said, ‘We can’t see him for a month.’ Dr. Beitler said, ‘double-book him.’ And they did.”
“He’s phenomenal,” says Allison Del Medico, a 30-year-old mother who was diagnosed with a rare salivary gland tumor last year. “There aren’t words to describe how amazing he’s been.”
Beitler emails her with MRI results and then calls to make sure she understands the results, Allison says.
That said, “he won’t let you stray,” Del Medico and others say.
“I asked if I could just have one glass of wine recently, and he said, ‘absolutely not.’”
“He told me, ‘this isn’t Burger King.’ You don’t get to have it your way,’” another patient, Jim Deweerth, chimes in.
Another patient recalls how Beitler and Scott Kono met him in the emergency room one Monday at 2 a.m.
Beitler stresses that everyone on the head and neck cancer team has the same dedication. He quotes from a former mentor that it is not only a duty to serve others but also a joy.
“It’s a great way to spend your life,” says Beitler.
As more patients than ever survive cancer, Winship’s Survivorship Program helps them thrive as they make the transition to the next phase of their lives.
By Diane Ross | Photography by Jack KearseJan. 2, 2009. That’s when the journey began for Shawn Ware, her husband Albert, daughter Demitria, son Jalen, and mother Eva Freeman.
A little lump she felt while in the shower took the family on the odyssey that is breast cancer. Ware had a lumpectomy and treatment with radiation therapy and chemotherapy.

“You know those side effects that you see in fine print? I had all those and more,” she says, somehow able to laugh about them now. “I didn’t know that your eyelashes act as windshield wipers, and when I lost mine, I had to wear glasses just to keep things from getting in my eyes.”
Ware triumphed. “I was ready to conquer the world after my last round of radiation,” she says. And three years later, she is considered a survivor and a reason for celebration.
“Cancer, it stinks,” says Ware, the general manager of Blomeyer Health Fitness Center at Emory. “But you do change. You certainly learn to appreciate the good and not let the little things bother you any more.”
Like millions of other Americans, Ware is part of a growing trend—more people than ever are surviving cancer. In just six years, the number of cancer survivors has jumped by almost 20 percent, according to the Centers for Disease Control and Prevention and the National Cancer Institute—11.7 million in 2007, up from 9.8 million in 2001, the most recent years available.
The good news comes with some challenges, however. As cancer treatment has become more successful, survivors —and their caregivers and providers—have learned that there is a cost to surviving.
Survivors of all types of cancer can face myriad physical issues. Treatment itself can be so hard on the body that survivors sometimes suffer chronic pain, heart problems, depression, sexual dysfunction, and a mental fogginess dubbed “chemo brain.”
They also are at heightened risk for recurrence and secondary cancers.
Physical problems arise within individual cancer groups. For example, head and neck cancer patients often have trouble swallowing and lose their sense of taste. Breast cancer patients must deal with the changes that come as a result of a lumpectomy or mastectomy and reconstruction.
In addition, family and relationship problems may arise as all in a survivor’s relationship network struggle to adjust to cancer and life after cancer. Emotional challenges abound, from sadness, fear, and anger to serious depression. Fatigue is common.

“We are now defining a ‘new normal’ for these patients. There can be longterm after-effects when treated for cancer, and we are finding ways to improve their quality of life while providing guidance on strategies for dealing with these after-effects.”
-Joan Giblin, director of Winship’s new Survivorship Program.“Long-term survivorship starts on the day treatment ends,” says nurse practitioner Joan Giblin, the director of Winship’s new Survivorship Program. “You’re actively doing something during treatment, but when treatment ends, many patients tell us they feel like they have been set adrift without a clear course. Our survivorship program is trying to bridge that gap and provide survivors with tools for these difficult times.”
Giblin says that some survivors respond by isolating themselves. Still others “jump right back into their old lives or try to adjust to a new life by adapting to any after-affects they may still be experiencing.”
Winship Cancer Institute is helping survivors deal not only with the late physical effects of cancer but also with the psychological and social issues that are part of surviving.

“We are now defining a ‘new normal’ for these patients,” says Giblin. “There can be long-term after-effects when treated for cancer, and we are finding ways to improve their quality of life while providing guidance on strategies for dealing with these after-effects.”
The Winship Survivorship Program officially started in November, 2011. Already more than 10 Winship survivorship “clinics” are being offered, focusing on survivors of 10 different cancer categories. The program holds workshops on such vital topics as nutrition, preventing lymphedema, how to talk to children about cancer, spirituality and pet therapy. Workshops have been held on sexuality and also on fatigue. In May, Winship announced its collaboration with the YMCA of Metro Atlanta for a special exercise program for cancer survivors. A unique collaboration, Winship at the Y was Giblin’s brainchild. She is at the hub of a very extensive interdisciplinary wheel that involves specialists from a wide range of treatment areas, including nutrition, pain management, and psychiatry to help survivors thrive.
Leukemia survivor Dick Bowley, of Peachtree City, is doing so well he recently was able to undergo hip replacement surgery, and he has published a book on his experiences with cancer to help other survivors deal with the issues they may face.

“We have to change how we look at cancer patients,” Giblin says. “Many cancers are not curable in a conventional sense, but the improvement in the quality and quantity of life needs to be our priority. Much as we view diabetes as a chronic condition, we must look at many cancers in the same way.”
Head and neck cancer survivor Barry Elson, 70, had difficulty swallowing after his treatment. Elson, who was first diagnosed in 2003, had an esophageal dilation last year to improve his ability to swallow.
“I think in the press of your day-today survivorship, you forget to ask what (the treatment) might do to your longterm quality of life,” Elson says.
Ware found that exercise has not only helped her gain physical strength but also has helped her mental outlook. Ware was able to exercise throughout
most of her treatment, even as ill as she was. Now, her worst worry is fatigue. But that doesn’t slow her down. In her job as fitness manager at Blomeyer, she conducts “boot camp” training sessions and teaches other classes.
Winship is also helping survivors thrive by providing support services to help survivors cope with employment and insurance issues that arise as a result of their cancer.
“After treatment,” Giblin says, “patients tend to not be able to work as long, and they don’t have the stamina they used to have.” In addition, there can be stigma in the workplace against a cancer survivor, which in times of layoffs, can result in their loss of employment and consequently, loss of benefits.
“It’s the people who can’t afford to lose their jobs who do,” she says. And even in cases where survivors
keep their insurance benefits, they might find a lack of integrated care as they celebrate more birthdays.
Paper records are lost through the years, hospitals and oncology offices change and primary care physicians— who don’t have experience in oncology —aren’t prepared or educated to provide the ongoing care cancer survivors need.
Elson says he fared well—a result, in part, of diligent Winship physicians Amy Chen and Dong Moon Shin, and the nursing staff—including Giblin.
Despite the side effects she faced during treatment, Ware says she has grown from her cancer experience. It makes her a stronger survivor, she says, and also more hopeful, optimistic, and motivated.
“It’s almost motivated me to do more,” she says. “It really helps me to live day by day. You make every day everlasting.”
The “divide and conquer” approach to cancer therapy has been increasingly prominent in recent years. This strategy identifies drugs that are particularly effective against tumors carrying certain growth-driving mutations.

Two recent examples include crizotinib, approved by the FDA for non-small-lung cancer with a translocation in the ALK gene, and vemurafenib, approved for melanomas with mutations in the B-raf gene.
One inevitable drawback: if someone’s tumor doesn’t carry the specified mutation, these drugs can’t help. The mutation that makes a tumor vulnerable to crizotinib is found in only a small percentage of lung cancers.
So when designing a research program in lung cancer, why not go after the targets that cause the most disease?
That’s the rationale behind Winship researchers’ hunt for ways to attack the worst of the worst: lung cancers with mutations in K-Ras, a gene that has long been considered “undruggable.”
Lung cancers with mutations in K-Ras, which make up around a quarter of all nonsmall cell lung cancers (NSCLC), are known to be resistant to both chemotherapy and newer drugs. In the 1980s, the K-Ras gene was one of the first oncogenes identified, by virtue of its presence in a virus that causes cancer in rats. Basic scientists have examined ways to inhibit the effects of mutated K-Ras in cancer cells, without much success.
Suresh Ramalingam, Winship’s chief of thoracic oncology and director of medical oncology, says K-Ras and another frequently mutated gene in lung cancer, LKB-1, are at the center of Winship’s strategy for lung cancer research. That strategy has been identified as a major focus for the Emory Lung Cancer NCI- funded program project grant entitled “Targeting Cell Signaling in Lung Cancer to Enhance Therapeutic Efficacy.” Winship’s deputy director, Fadlo R. Khuri, and Haian Fu, both of whom have devoted a number of years to identifying cancers with K-Ras mutations, lead that application, and targeting K-Ras is a major focus of a project in the grant co-led by Ramalingam and Yuhong Du.
“The significance of K-Ras mutations, as far as poor outcomes with chemotherapy with NSCLC, has been well known, ” Ramalingam says. “The field has been looking for a way to target K-Ras for a long time. You can think of this as a big white elephant sitting in the room, demanding attention.”
Now research led by Winship researchers has provided leads for two strategies that may help doctors capture this elusive target. Both involve anticancer agents that are already in clinical trials.
One of the leads involves “death receptors,”
molecules on cells that act as built-in self-destruct buttons and halt the progress of cells that could become cancerous. Finding a way to push these buttons on cancer cells has been an appealing goal, but clinical studies of antibodies that target death receptors have been disappointing so far.
In a paper published in Journal of Biological Chemistry, Winship researcher Shi-Yong Sun and colleagues have shown that antibodies that target death receptors are more effective against cells with mutations in K-Ras. The mutations cause the cells to put more death receptors on their surfaces, making the cells more vulnerable.
Another promising strategy involves observations from a recent trial led by Ramalingam. The study was testing a combination of erlotinib, an FDA-approved drug for non-small cell lung cancer, and an antibody against insulin-like growth factor receptor (IGF-1R), an experimental drug. Though the combination did not prove successful for the entire study population, the investigators discovered that those with K-Ras mutations seemed to do better.

“In this study, the presence of a K-Ras mutation was associated with a poor outcome with erlotinib alone but predicted a favorable outcome with the combination,” Ramalingam says. “This suggests that future trials of this combination with IGF-1R targeted agents should focus on patients whose tumors carry the K-Ras mutation.”
He adds that the emphasis on K-Ras is part of a larger effort to tailor lung cancer therapy to each patient’s disease. Winship has been participating in research studies, such as the Lung Cancer Mutation Consortium, a 14-center National Cancer Institute-supported study where each patient’s tumor DNA is checked for a panel of common mutations, including K-Ras. That information is then used to direct their treatment, either to known targeted therapies or to clinical trials of new therapies.
“Our eventual goal is to be able to offer every patient the best therapeutic options, as determined by genetic analysis of the tumor, whether they have a K-Ras mutation or not,” Ramalingam says.
Melanie Seymore, left, received such great care from surgeon Toncred Styblo, right, that Seymore and her husband asked wedding guests to donate to Winship rather than give presents to the newlyweds.
Melanie Seymore had plans to get married. But instead of getting married in 2010, she was diagnosed with breast cancer.
“It was pretty shocking, because I was considered the healthy one,” she says. “I worked out, I ate vegetables. I wasn’t expecting it.” And yet despite a cancer diagnosis, treatment, surgery, the tragic loss of her fiance’s parents, Melanie and now-husband Dan Mowery decided she had to give back to Winship. In an extraordinary gesture of generosity, the couple asked that guests for their long-awaited wedding make donations to Winship instead of buying traditional wedding gifts for the couple.
“Melanie is just an exceptional person,” says Toncred Styblo, the Winship surgical oncologist who performed Melanie’s lumpectomy. “She is very strong, very caring. We were so happy to be able to see her get married, see her wedding photos, and

share in that time with her. And to think that she donated her wedding gifts to Winship was really more than we could imagine. She’s just amazing.”
Melanie knew that Dan was a good catch as soon as she met him in 2007. Both Chicago natives, they immediately hit it off, sharing memories of their hometown.
The couple became engaged in 2008. For a 40-something-yearold career woman who had waited decades for her soul mate, her engagement was a joy not only for her but also for all those who knew her and cared about her and husband-to-be.
After waiting so long to find Dan, Melanie knew she wanted a fairy-tale wedding—family, friends, and a beautiful gown. They planned to marry in 2010. Dan and Melanie were trying to sell one of their houses to prepare for moving into one house after they married. An uncooperative housing market interfered.
Then, in July, 2010, Dan’s parents were killed in a car crash.
“They went out to get his mother a haircut,” Melanie says. Dan’s father lost control of their car, and it slammed into a brick wall. Dan’s father died within 24 hours of the accident; his mother died a few days later. The grief of the sudden loss was especially difficult, Melanie says. There was no way the couple could consider rejoicing in a wedding when their hearts were breaking from the loss of Dan’s parents.
Three months later, Melanie was diagnosed with breast cancer. She knew she would have to postpone the wedding.
“I didn’t want to be a bald-headed bride,” Melanie says. “I had seen far too many TV wedding shows with brides trying dresses on with veils. I wasn’t going to give that up.”

Melanie had a type of breast cancer called triple negative. It is called triple negative because it tests negative for three different kinds of receptors on the surface of breast cancer cells. Researchers have developed effective treatment that targets those receptors. Because triple negative breast cancer does not have those receptors, it is less vulnerable to those drugs. It can be a very challenging cancer to treat.
The day she received her diagnosis, she recalls feeling very comforted by the kindness of surgeon Styblo.
“She held my hand and hugged me and assured me that everything was going to be OK,” Melanie says. “I felt very fortunate I was there.”
Melanie had chemotherapy, a lumpectomy, and radiation. Amelia Zelnak, whom she says was “absolutely wonderful,” was her medical oncologist.
Dan was beside her every step, she says, even being acknowledged by the American Cancer Society as one of three top caregivers of the year.
“In fact, he went with me to every single chemo visit, using up all his vacation days,” Melanie says. “He acted like my valet.”
Dan says it was very hard watching Melanie go through treatment. When he was a teenager, his father was diagnosed with cancer.
“I can remember the moment my mother told me,” he says. “That memory was seared into my heart.”
her find answers—and helped her stay calm. “Every time I’d read something about triple negative, I’d email Heather. I would get so upset. Heather said I needed to get offline.”
Heather also helped lift her spirits. “When you go through breast cancer treatment, you don’t feel pretty any more,” Melanie says. “It’s really a hit to your self-esteem.”
By the summer of 2011, things had begun to change. Her hair was growing back. She had energy. She was feeling good and hoping that she and Dan could finally plan their wedding. Their chance arrived on Oct. 29, 2011. They finally married. As they were planning their wedding, they needed to decide where to register for gifts that their friends and family would be interested in buying for them.
“Everybody kept saying, ‘where are you going to register?’” Melanie recalls.
“I would just want every single woman who goes through cancer to know there’s a light at the end of the tunnel and that there can still be a fairy tale ending.” —Melanie
SeymoreGoing through cancer treatment with Melanie almost broke his heart.
“You hate to see anyone suffer, but when it’s someone you love so much, someone like Melanie…it was tough,” he remembers.
He did everything he possibly could to help his fiancée, and he was glad to be able to do so, he says. He credits great co-workers and a network of friends who were there for him so he could be there for Melanie.
“Sometimes these kinds of things tear a couple apart,” he says. “But not us, thank goodness. It made us stronger.”
Melanie also credits breast cancer nurse navigator Heather Pinkerton, who helped
And after all the trials and heartbreaks the couple had been through, they thought of other people at one of the times in people’s lives when the spotlight rightly should fall on them.
“And I thought, ‘why don’t we just ask people to donate to Winship?’
The choice was obvious, Melanie says. To date, their guests have donated more than $6,000 to Winship.
“I would just want every single woman who goes through cancer to know there’s a light at the end of the tunnel,” says Melanie, “and that there can still be a fairy tale ending.”
Do you have a story of heroism or kindness about a patient, doctor, nurse, or other staffer at Winship that you’d like to share? Tell us about it for The Winship Way. Send your story to: virginia.l.anderson@emory.edu, or call at 404-778-4580.
When the Atlanta Board of Education needed the best professional ethicist in town to head its newly formed ethics panel, it actually was able to engage one of the best ethicists in the nation. Rebecca Pentz, professor of hematology and medical oncology in research ethics, Winship Cancer Institute researcher, and one of the nation’s leading ethicists, was named chair of the ethics commission of the Atlanta Board of Education.

Pentz, who holds a doctorate in philosophy from the University of California at Irvine, is a widely published investigator and author in cancer-related bioethics. Her research focus is empirical ethics research on such issues as informed consent, phase 1 research (first use of a drug in humans), children with cancer, and genetic confidentiality, as well as assisting other researchers in making their clinical trials more ethically sound.
“I’m very honored to be named chair of this commission,” says Pentz of her Board of Education duties. “My kids all went to public school, and I believe public schools are a wonderful way for people to receive an education. I am actually quite flabbergasted that I was elected chair from among the distinguished professionals who serve on this commission. I’m thrilled to be able to do it.”
And Winship is very honored to have Pentz, says Deputy Director Fadlo R. Khuri.
“Becky Pentz is one of the finest clinical and research ethicists in medicine,” Khuri says. “She formed the clinical ethics program at MD Anderson, and since joining Emory University and the Winship Cancer Institute, she has developed one of the premier research ethics programs in cancer. She has done pioneering work on research in vulnerable populations and patient perceptions of
Winship is one of the few cancer centers in the country to have a fulltime ethicist dedicated to research and clinical trials. Now, Rebecca Pentz is helping another important organization as it charts its ethical course.Courtesy of Shannon Bell Photography
their participation in cancer clinical trials. I extend my congratulations to the City of Atlanta on appointing by far the best qualified and best person that they will ever have on their ethics board.”
While medical ethicists have become more visible in large hospitals as an adjunct to their clinical programs, Winship is unusual in that it has a full–time ethicist on faculty overseeing its research programs.
“Winship really wanted to go the extra mile,” Pentz says. “It’s very unusual to have an ethicist embedded in a research program.”
What role does an ethicist play in medical research? Think Henrietta Lacks and her immortalized cells, or the infamous Tuskegee studies involving syphilis. While medical research has evolved and learned from such experiences, the field of bioethics has become more and more important as the medical terrain has become more complicated. Health care consumers can barely keep up with the complicated language of their diagnosis, let alone know how to assess whether they want to enroll in a trial or share their medical information—not to mention actual parts of themselves such as tissue samples—with researchers.
Or think about the people on the outskirts of medicine. The project dearest to Pentz’s heart is designing an intervention to help the siblings of children with cancer. “There is no one in health care really assigned to these children. Yet they suffer too.”
One of Winship’s greatest strengths—its drug development program —can be an area of confusion for patients, Pentz explains. What’s a phase 1 trial, and how does it differ from phase 2 or 3 trials?
“This is why it’s so hard for patients,” says Pentz. “The phases are all different, and patients need to know the difference, so we spend a lot of time designing ways to help patients better understand what is involved when they’re giving consent. One of the things we’re doing right now is to flag all research procedures that are extra and not necessary to take care of the patient. Simply, we are trying to make it really, really, really clear what’s going on in a trial.”
Some of it involves “pretty complicated concepts,” Pentz says, such as understanding that the goal of a patient enrolling in a phase 1 trial is to assist researchers in determining the safest drug dose for human use. Because patients almost always hope that their cancer will be cured, it is morally and ethically essential to completely inform them about the purpose of enrolling in a phase 1 trial. It is Pentz’s and fellow Louisa Wall’s role to help patients understand that.
“Phases 1 trials are about pioneering, innovative research,” Pentz says. “So we have to make that really clear, because you’d never offer patients a hope for a cure when you know the chances of that are small.”
That said, “We would never try a drug without it having the possibility of helping patients,” Pentz explains. But she and others involved in the trial must work as hard as possible to ensure that patients enter trials with realistic expectations.
Pentz says she finds her work “fascinating.”
“It’s such a great place to work,” she says. “It’s the most collegial place I’ve ever worked—the chemists, biologists, clinicians, statisticians, and even the ethicist, all meet together regularly. This kind of collaboration just doesn’t happen at other places.”
You may have seen Rebecca “Becky” Pentz around the halls and in the labs at Winship, but you may not know these things about her—she likes singers Adele and Zac Brown, and she is married to a Presbyterian minister. She wanted, as have many people, to become a doctor “until I hit organic chemistry.” She never lost her interest in medicine and was able to blend it with her fascination with ethics. Read on for more things about Pentz.
Birthplace: Seattle
Education: BA, Philosophy, Pomona College; MA, Philosophy, Bryn Mawr; PhD, Philosphy, University of California, Irvine, 1979.
Family: Husband, Vic, Pastor of Peachtree Presbyterian Church; three married daughters, Sarah, Jessica, and Amy; three grandchildren(pictured at left).
Favorite music:
“I really like the Eagles, but I do have to admit I like Adele and Zac Brown.”
If I could switch jobs with anyone at Winship, it would be:
“Nobody! I have the best job at Winship. I think a lot of people wish they had my job!”
nity, we had 1,700 runners and walkers, and we raised more than $200,000. We also had a great time bringing together the community and raising awareness about the fight against cancer.”
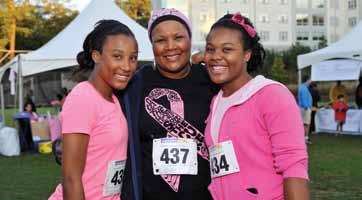
Curran himself also had a great time on the course—he won the 60–year–old–and–older category!
Race promoters across the state were amazed at the success of the race. First races often attract a few hundred runners and grow steadily over the years. The Winship Win the Fight 5K got off to such a fast start that those familiar with races predict the race will become one of the fall’s featured running events.
The inaugural Winship Win the Fight 5K in October, 2011 was a phenomenal success, exceeding initial goals of both runners and funds raised by a factor of two! Now, join Winship for the second annual Winship Win the Fight 5K to walk or run on Saturday, Oct. 13, 2012!
“We hoped to have 700 participants and to raise $100,000 to support Winship’s research mission,” says Walter J. Curran, Jr., executive director of Winship and chief promoter of the 5K. “Through the amazing support of our friends and the commu-
Advances in lung cancer research, a world-class multiple myeloma program, the Southeast’s largest head and neck cancer research effort, and a broad portfolio of cancer clinical trials are among the many reasons that Winship successfully renewed its National Cancer Institute (NCI) cancer center designation status.
Officials at Winship recently received formal notification from the NCI of its decision and also learned that nearly $8 million in funding from the NCI over the next five years has been allocated to support this designation.
“On behalf of all of Winship’s faculty and staff, we are grateful to the NCI for its continued support, and we appreciate that the reviewers recognized the outstanding
The fact that the race is a Peachtree Road Race qualifier no doubt helped attract a number of runners, but many of those who participated in the race for a reason other than to qualify for Peachtree said they derived a great deal of personal satisfaction from the race. The Rev. Joseph Peek, a Roman Catholic priest who walked the race, used the experience as a way to raise money for Winship and to help those who may have been too sick to participate. Peek, a leukemia survivor, has struggled for years with graft-versus-host disease and for many months was too weak to walk. The inactivity was challenging for the former Navy rescue swimmer. The Winship Win the Fight 5K became a victory lap of sorts for the him. And his team—Patient Endurance—raised more than $2,000.
The course will start again at McDonough Field on the Emory campus and wend its way through the beautiful Druid Hills neighborhood. Registration has begun, and you can start assembling your team—and fundraising—now! Mark the date, join the race, and help Win the Fight!
nature of our research,” says Walter J. Curran, Jr., Winship’s executive director.
A panel of 23 NCI-appointed cancer research experts reviewed Winship’s application for this renewal and scored Winship at the “outstanding” level. Only 57 other centers nationwide that provide care to adult cancer patients hold this NCI cancer center designation, and Winship is the first and only NCI-designated cancer center in Georgia.
Curran stresses that this designation is at once an emblem of trust and a responsibility that Winship takes seriously.
“The designation is not about bragging rights,” Curran says. “It’s about a standard of research and care that patients have learned to trust.”

Winship’s second gala was held April 14 at the Piedmont Driving Club, and it raised more than $600,000 for research programs at Winship. It was an elegant evening of dinner, dancing, and celebrating achievements in the fight against cancer. With HOPE as its theme, the signature event, organized by Friends of Winship, sold out at the patron level. Gala co-
chairs and Friends founding members Ann Hastings and Leslie Wierman and their dedicated committee did an outstanding job organizing this event, which recognized Mary and John F. Brock, CEO of CocaCola Enterprises, Inc., as honorary chairs. Thanks to everyone who helped make the Winship Gala 2012 a tremendous success!

Receiving a cancer diagnosis not only leads to the question of “How will I beat this disease?”, but also “How do I afford it?” Jim Hankins, social services director of Winship, aimed to answer the latter concern when he established the Winship Patient Assistance Fund in 2002.
The funds come from a variety of sources, with donations often varying in size, says Hankins. For example, Paint Georgia Pink last December made a very generous donation to help breast cancer patients with financial need. Paint Georgia Pink, which raises money in memory of Randi Passoff, holds especially novel fundraisers, such as its Pink Pirate’s Ball in February, to help breast cancer patients with financial need.
The fund also receives support from other fundraising endeavors, both from patients as well as from the Winship community. “Everyone’s coming together to make this happen,” says Hankins. The amount of money available depends on ”what’s in the account,” explains Hankins. “When funds are available, we are able to assist.”
Because of the sluggish economy, more patients are facing financial challenges than they are when times are good, Hankins says. Thus, donations are especially appreciated.
Donations are accepted in form of cash or check made out to the Winship Patient Assistance Fund. Contact Mark Hughes at (404) 778-1288 for questions about donating.
Six yearS into their marriage, george Weaver lost his wife, allison, to breast cancer. She was only 37.
nearly 25 years later, Weaver has made Winship Cancer institute of emory University the beneficiary of a life insurance policy to support breast cancer research in allison’s honor. “We learned that when breast cancer hits young women, it is often very aggressive,” says Weaver, now remarried with two children. “i wanted to do something to fight this terrible disease that could take a young person in the prime of life and snatch her away.”
Learn how your estate gift can aid Winship in the fight against cancer. Call the office of gift Planning at 404.727.8875, email giftplanning@ emory.edu, or visit www.emory.edu/giftplanning.
