|
|
 |
||||
 
|
 |
||||
| In Isaac Asimov’s classic science fiction novel, Fantastic Voyage, a group of adventurers shrink themselves and travel through the human body in a miniature submarine with the mission of destroying a lifethreatening blood clot. Today, a team of Emory researchers, using the principles of nanotechnology, is creating miniscule, engineered semicon-ductor particles called “quantum dots” that can travel through the bloodstream, seek out tumor cells, and pinpoint hundreds of variations in their genes and pro-teins. In the future, these nanoparticles may serve as min-iature delivery vehicles, unloading packages of drugs that are just what the doctor ordered to attack a parti-cular type of tumor cell. Ever since the sequencing of the human genome, physicians and patients alike have envisioned the promising prospect of personalized cancer medicine. Combining a patient’s genetic profile with the unique characteristics of a particular tumor might yield just the right combination of therapies to target and treat an individual’s specific disease. The Emory nano-scientists believe this diminutive new technology could soon help fulfill the genome’s promise of individualized diagnosis, prognosis, and therapy. Landscape of a dot Nanoparticles, the basic building blocks of nanotech-nology, range from 1 to 100 nanometers on the nanoscale. One nanometer is one-billionth the size of a meter (about 100,000 times smaller than the width of a human hair), and a nano-metersized particle is about the size of three to four atoms. Although many biologic particles—including DNA, proteins, and genes—fall within the nanoscale range, the term “nanotechnology” generally is reserved for materials constructed by humans, one atom or molecule at a time. Nanoparticles on the lower end of the scale, ranging from about 1 to 3 nanometers, assume special “quantum” properties simply by virtue of their miniature size. For example, if you break a piece of candy into two pieces, each piece will still be sweet, but if you continue to break the candy until you reach quantum proportions, the smaller part will taste completely different and will have different properties. A piece of gold appears to be yellow, but broken down into nanoparticles, it appears to be red. Broken down even smaller, it appears to be blue. Shuming Nie, director of cancer nanotechnology at Emory’s Winship Cancer Institute and professor in the Coulter Department of Biomedical Engineering at Georgia Institute of Technology and Emory, is working to harness the unique properties of nanoparticles. One of the first scientists in the world to use nanotechnology for medical applications, Nie was recruited to Emory in 2002 with the help of the Georgia Cancer Coalition. While on the chemistry faculty at Indiana University, he began constructing semiconductor quantum dots with an eye toward cancer diagnosis and treatment. |
|||||
| These
nanoparticle “dots” combined cadmium with |
|||||
 |
|||||
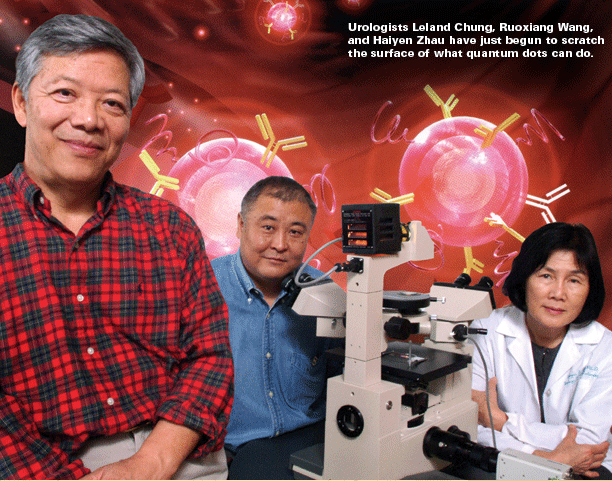
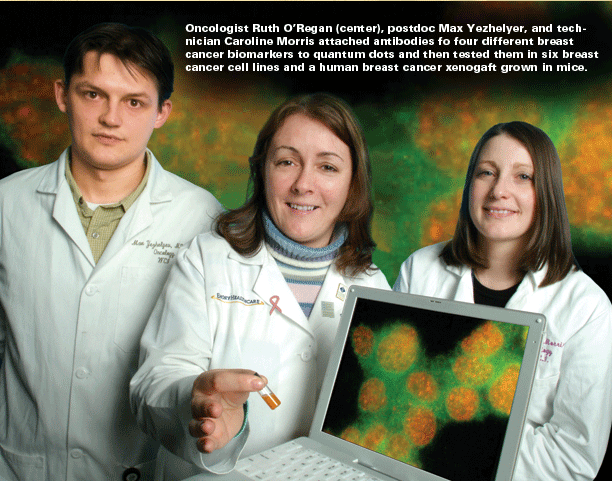
Zip code of a cancer cell The translational work of the grant already has far outreached expectations in the first few months, says Nie. Emory breast cancer oncologist Ruth O’Regan has attached antibodies for four different breast cancer biomarkers (estrogen receptor, progesterone receptor, her2-neu, and Ki-67) to quantum dots and then tested them in six breast cancer cell lines and a human breast cancer xenograft grown in mice. So far, her results correlate favorably with standard detection methods. The next step will be to test the dots in a bank of archived breast cancer specimens, followed by a clinical trial with current patients, comparing quantum dot detection to standard methods using eight breast cancer biomarkers. The current diagnostic technology of immunohistochemistry requires that pathologists look at only one biomarker at a time, using a different thin slice of stained tissue to examine each marker under the microscope. Each slice, reflecting a different section of the tumor, may miss potentially important information. “Because there are so many markers for breast cancer, we haven’t been able to apply them all,” says Chung. “We are just beginning to scratch the surface of what quantum dots can do. We keep talking about early diagnosis and treatment, but we don’t have a way to monitor that. In prostate cancer, this technology has dramatically improved the sensitivity and specificity of how we view tumors in mice. Clinically, we can see a tumor about 1 centimeter in size, which is 1 billion cells. Quantum dots allow us to see tumors with just 100 cells. “With quantum dots,” he explains, “we can recognize these markers quickly in one shot, which would be a tremendous advantage in treating patients. The molecular signature of a cancer cell could serve as its own zip code for optimal treatment. This ability to fill in the blanks could completely change medicine.” “Our whole rationale,” says O’Regan, “is to try to determine, using the quantum dots, what treatments are appropriate for patients so we can tailor the treatments. We give a lot of people chemotherapy, but we don’t know if they all need chemotherapy. We hope to find an expression pattern associated with response to certain therapies. This is just the tip of the iceberg.” Emory prostate pathologist and researcher Milton Datta is using archived tissue specimens from more than 700 prostate tumors from Emory patients to analyze genes and other biomarkers that might be used with quantum dots to help diagnose early cases of cancer. A recent study published in the New England Journal of Medicine showed that some men with low levels of prostate-specific antigen have cancer that goes undetected. Datta and his colleagues are studying the proteins and attached sugars shed by prostate tumors in the bloodstream. He hopes quantum dots may be able to measure changes in levels of these proteins and sugars to detect prostate cancer at an earlier stage. |
|||||
 |
|||||
Hurdles along the way Emory nanotechnologists have recently started a new initiative in nanotherapeutics—using nanoparticles to deliver drugs. Nie has packaged the commonly used cancer drug taxol into biodegradable nanospheres engineered to seek out head and neck cancer cells and release their drug contents at the tumor site. The technology has worked successfully in mice. By targeting the drug directly to the tumor, these smart nano-packages would create less toxicity for the rest of the body and deliver a direct hit to the tumor. Datta, Nie, and Chung soon will be testing the packaging tech-nique in mouse prostate tumors with the drug cyclopamine, which has been shown to block the prostate growth-enhancing gene, sonic hedgehog. The highest hurdle remaining before nanotyping can be used for diagnosis and prognosis in patients is the potential toxicity of the quantum dots. Although cadmium on its own is highly toxic, the cadmium selenide compound in theory shouldn’t be, says Nie, and the special polymer coating encap-sulating the quantum dots is designed to protect against any toxicity. Although no toxicity has yet been discovered with the cadmium-selenide dots in cells or tissues, an acute toxicity study still needs to be done, he says. Although the unknown toxicity currently restricts physicians’ use of quantum dots to treat patients, researchers already can use “their multiplexing ability to monitor hundreds of markers in tissue samples,” according to Chung.For Datta, “One of the most exciting things about our work in nanotechnology is that it brings together scientists from medicinal chemistry, nanotechnology, engineering, biology, and bioinformatics along with physicians, all working around an exciting technology and focusing on diseases they care about and want to cure.” The fantastic voyage is just beginning. 
Holly Korschun is director of science communications for the Woodruff Health Sciences Center at Emory. |
|||||
TOP past issues . contact us . home give a gift . som home |
|||||
| Copyright © Emory University, 2004-2005. All Rights Reserved | |||||
 color
variations. In 1998, he and a graduate student published a groundbreaking
paper in Science that described “quantum dot bioconjugates”—
luminescent nanoparticles that could be chemically bound to biologic
particles such as anti-bodies, peptides, proteins, or DNA. These bioconjugated
dots could be engineered to detect other molecules, such as biomarkers
on the surface of cancer cells. The paper became “News of the
Week” at both Science and Chemical Engineering News.
The cover of Nature Biotechnology described Nie’s method
of encapsulating various sizes and colors of quantum dots within microbeads.
Nie’s own face lights up as he enthusiastically describes the
promise of nanotechnology in medicine.
color
variations. In 1998, he and a graduate student published a groundbreaking
paper in Science that described “quantum dot bioconjugates”—
luminescent nanoparticles that could be chemically bound to biologic
particles such as anti-bodies, peptides, proteins, or DNA. These bioconjugated
dots could be engineered to detect other molecules, such as biomarkers
on the surface of cancer cells. The paper became “News of the
Week” at both Science and Chemical Engineering News.
The cover of Nature Biotechnology described Nie’s method
of encapsulating various sizes and colors of quantum dots within microbeads.
Nie’s own face lights up as he enthusiastically describes the
promise of nanotechnology in medicine.